A non-ionic water-dispersible surfactant for lipid-based formulations to solubilize and increase oral bioavailability of poorly water-soluble APIs. Self-emulsifies in aqueous media forming a fine dispersion, i.e., microemulsion (SMEDDS).
Names & synonyms
| USP NF Name | Mixture of Lauroyl Polyoxyl-32 glycerides and PEG 6000 |
|---|---|
| EP Name | Mixture of Lauroyl Macrogol-32 glycerides and PEG 6000 |
| UNII Code (FDA) | H5ZC52369M 30IQX730WE |
| Preferred Substance Name (FDA) | LAUROYL PEG-32 GLYCERIDES POLYETHYLENE GLYCOL 6000 |
| Handbook of Pharmaceutical Excipients | Polyoxylglycerides Polyethylene glycol |
| Chemical description | Consists of a small fraction of mono, di- and triglycerides and mainly PEG-32 (MW 1500) mono- and diesters of lauric acid (C12) and of PEG-150 (MW 6000) |
Key characteristics
| Product form | Pellets |
|---|---|
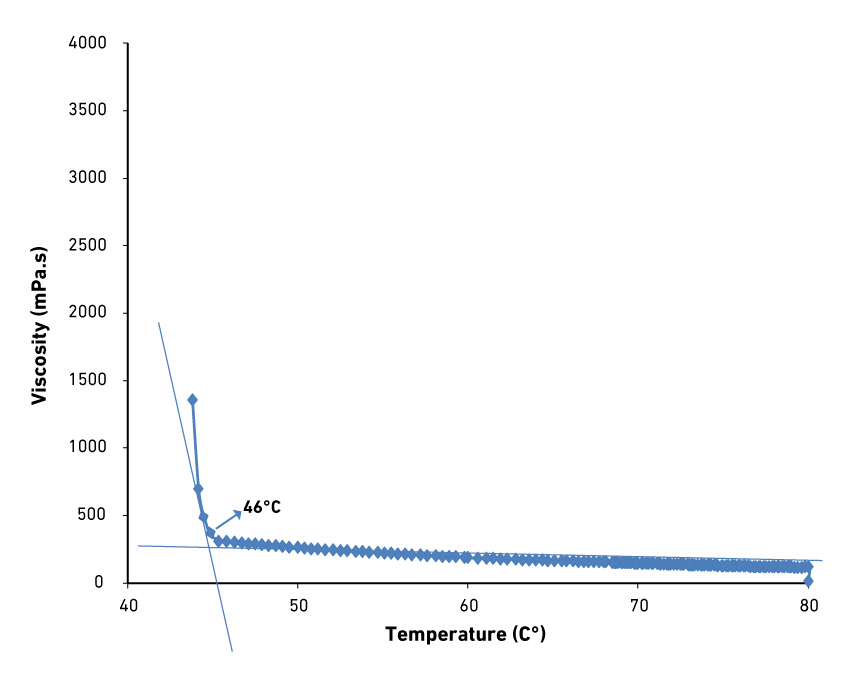
| Melting range (°C) | 57 - 62 |
| HLB (PIT method) | 14 ± 1 |
| CMC (mg/L, 25°C) | 40 |
Main functionalities
Solubilizer for poorly-soluble APIs and bioavailability enhancer. Single excipient formulation system: self-emulsifies in aqueous fluid into microemulsion—LFCS Type III (SMEDDS). Safety of use is inferred by extensive toxicological evaluations and precedence of use in approved pharmaceutical products.
Main formulation technologies
Self-emulsifying lipid formulation for oral applications.
Resources
Technical documentation
Associated content
Gelucire® 44/14
- Bioenhancer
- Self emulsifying drug delivery system
- Solubilizer

Excipients for solubility and bioavailability enhancement
Poor solubility, poor permeability, and pre-systemic elimination are factors that…