Lipid-based formulations
Lipid-based formulations are a powerful tool to increase oral bioavailability of poorly water-soluble or poorly permeable drugs. They can be used from early-stage development to market, significantly reducing the development time. Consisting in a mixture of excipients they are easy to formulate, scale-up and produce.
Developing successful lipid-based formulations step by step
An optimized lipid-based formulation enables solubilization of the entire therapeutic dose and maintains the drug in solubilized state throughout the digestion process. It is developed in 3 key steps:
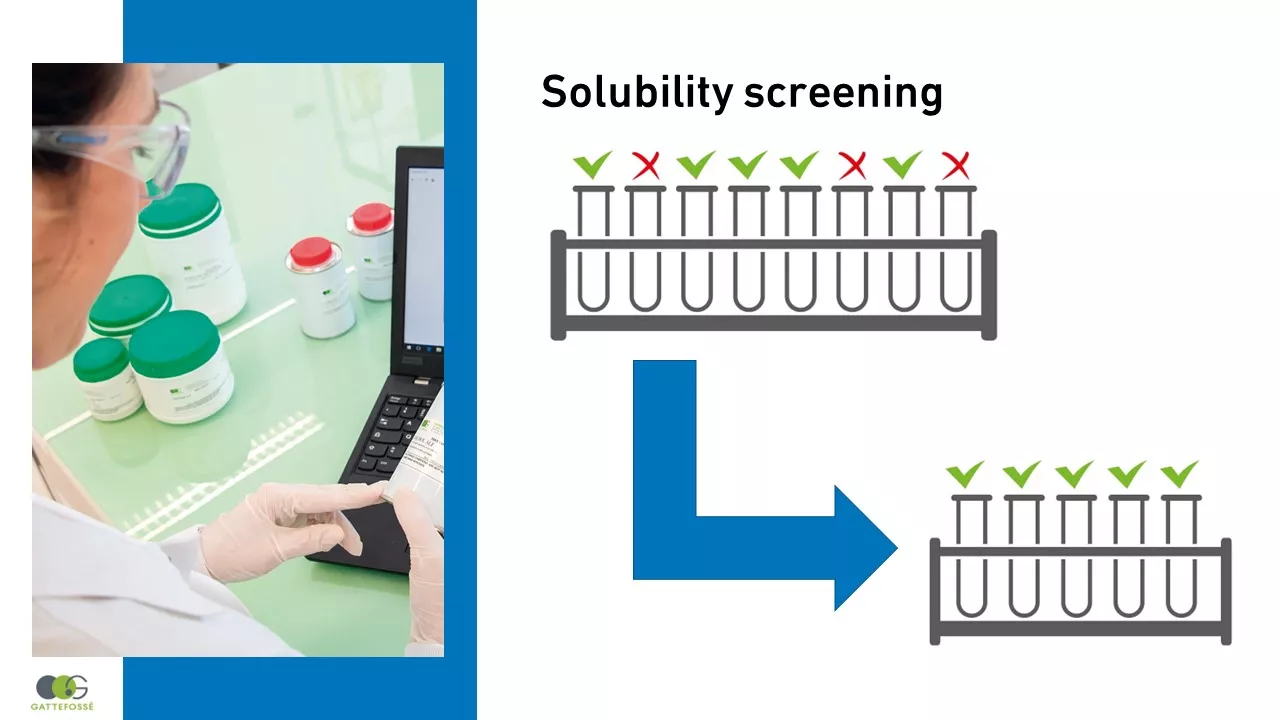
First, measure the saturation solubility of your drug in a range of excipients: oils, surfactants, and solvents.
Select the excipients that can solubilize your API.

Second, test the miscibility of binary mixtures of excipients and their dispersibility in water.
Select excipients that are miscible and dispersible in aqueous solution.

Third, perform the in vitro digestion test to evaluate the ability of the formulation to maintain the drug in solubilized state throughout the digestion test.
Go for in vivo studies with the best formulations!
To assist in lipid-based formulation development, we have produced tools to guide at each step:
Exploring the taxonomy, synonyms, and acronyms of lipid-based formulations
The literature on lipid-based formulations is abundant and many different words are used to describe lipid-based formulations. Let’s explore them and understand the similarity and differences.
Self-emulsifying lipid formulations, or self-emulsifying drug delivery systems, form emulsions upon contact with aqueous fluids without the need for mechanical or thermal energy. The size of the dispersion depends on the composition of excipients in the formulation:
SMEDDS: self micro emulsifying drug delivery systems
SNEDDS: self nano emulsifying drug delivery systems

LFCS: Lipid Formulation Classification System is used to facilitate the identification of the lipid-based formulations, based on the composition, the size of the droplets generated after the dispersion of the formulation in aqueous media, and the impact of natural lipid digestion process on the in vivo performance of the formulation classes (Type I, II, III or IV).