Excipients for solubility and bioavailability enhancement
Poor solubility, poor permeability, and pre-systemic elimination are factors that can limit absorption of some drugs. Lipid excipients have the capability to overcome these hurdles and enhance oral bioavailability through different mechanisms.
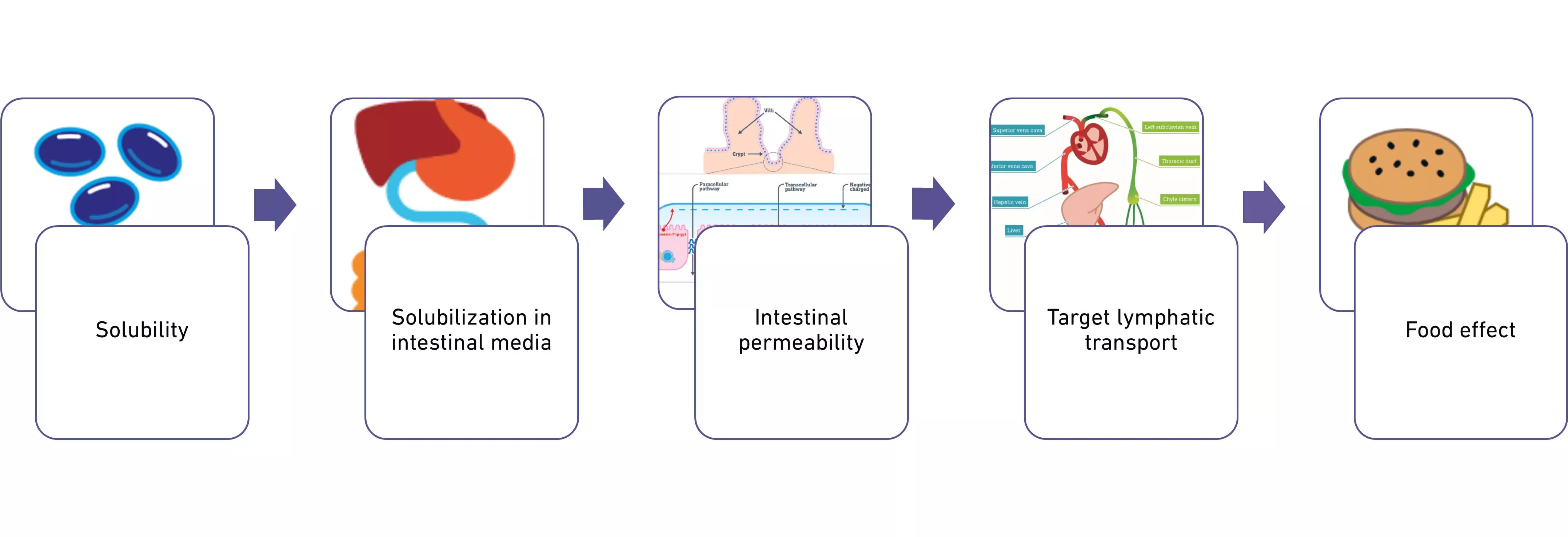
Lipid excipients offer a unique combination of benefits
Increase drug solubility
Poorly water-soluble drugs are generally soluble in lipid excipients.
Maintain drug solubilization throughout digestion
Upon action of enzymes and bile salts, the lipid-based formulation is digested and transformed in a series of colloidal structures: vesicles, mixed micelles, and crystalline lipid phases. They contribute to maintaining the drug in a solubilized state throughout the digestion process. Ultimately, fatty acids, monoglycerides and drug partition out of the micelles and are absorbed.
Increase intestinal permeability
Medium-chain fatty acids (C8-C10) are known to facilitate intestinal absorption of poorly permeable drugs via:
-
Transcellular uptake due to a membrane fluidization effect
-
Paracellular uptake due to the reversible opening of tight junctions.
Target lymphatic transport
Two prerequisites to promote lymphatic absorption:
-
As a general rule, the drug should be highly lipophilic (Log P > 5) and soluble in triglycerides (>50 mg/g)
-
The formulation must contain unsaturated long-chain fatty acids (C16-C18:1, C18:2) known to facilitate lymphatic uptake via assembly of drug with lipoproteins in the chylomicrons.
Mitigate food effect
Ingestion of a lipid-based formulation is sufficient to trigger the release of bile and lipases, in the same manner and extent as it occurs with a fat-containing meal. The difference between fasted and fed state is minimized and food effect can be reduced or cancelled.
A full range of excipients to enhance oral bioavailability
Our range of functional excipients includes oils, low HLB surfactants, high HLB surfactants and solvents. They can be combined to formulate Type I, II, III or IV lipid-based formulations.
Oral bioenhancers
Discover how Gattefossé excipients used in lipid-based formulations can enhance oral bioavailability of drugs.
-
Brochure Gattefosse Lipid-based formulations Bio-enhancers by nature pdf - 5 Mo -
-
Gattefosse brochure_Labrasol ALF, premium bioenhancer pdf - 5 Mo -
-
Gattefosse interactive brochure - Boost your drug bioavailability with Labrafac MC60 pdf - 4 Mo -
-
Brochure_Gelucire 44 14_ Self-emulsifying excipient for solubility and oral bioavailability enhancement pdf - 2 Mo -
-
Gattefosse brochure Gelucire® 48/16 pellets for solubility and oral bioavailability enhancement (En) pdf - 2 Mo -
-
Gattefosse brochure Maisine CC a pharmaceutical oil for solubility and oral bioavailability enhancement (En) pdf - 1 Mo -
-
Gattefosse brochure Lipid excipients for cannabinoid drug products pdf - 14 Mo -